July, 2002
Data Sets
Diauxic Growth Experimental Design and Data
H2O2
Treatment Experimental Design and Data
Minimal Glucose
Medium Aerobic and Anaerobic Growth Curve Experimental Design and Data
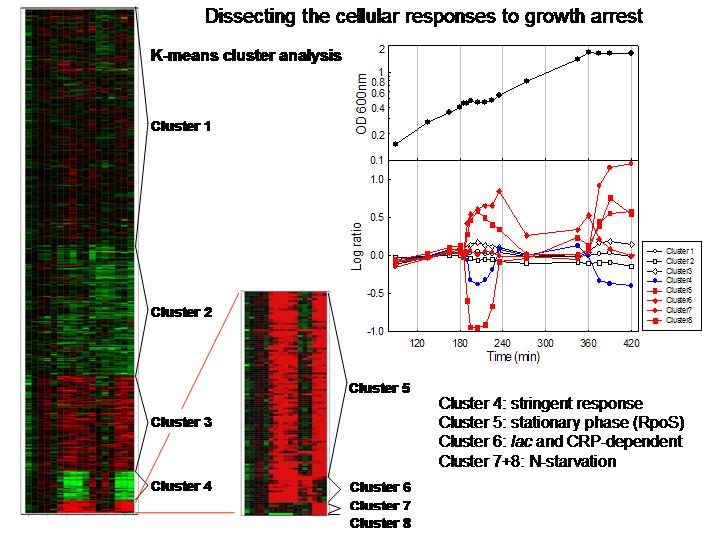
Project summary.
When conditions cause bacterial growth to stop, extensive reprogramming
of physiology and gene expression allows for the cell’s survival. We
used whole-genome DNA arrays to determine the system response in E.
coli cells experiencing transient growth arrest caused by glucose-lactose
diauxie and H2O2-treatment,
and also entry into stationary phase. The results show that growth arrested
cells induce stringent control of several gene systems. The vast majority
of genes encoding the transcription and translation apparatus immediately
down-regulate, followed by a global return to steady-state when growth
resumes. Approximately one-half of the amino acid biosynthesis genes down-regulate
during growth arrest, with the notable exception of the
his operon which transiently up-regulates
in the diauxie experiment. Nucleotide biosynthesis down-regulates,
a result that is again consistent with the stringent response. Likewise,
aerobic metabolism down-regulates during growth arrest, and the results
led us to suggest a model for stringent control of the ArcA
regulon. The stationary phase stress response
fully induces during growth arrest, whether transient or permanent, in
a manner consistent with known mechanisms related to stringent control.
Cells similarly induce the addiction module antitoxin and toxin genes
during growth arrest; the latter are known to inhibit translation and
DNA replication. The results indicate that, in all aspects of the response,
cells do not distinguish between transient and potentially permanent growth
arrest (stationary phase). We introduce an expanded model for the stringent
response that integrates induction of stationary phase survival genes
and inhibition of transcription, translation, and DNA replication. Central
to the model is the reprogramming of transcription by ppGpp,
which provides for the cell’s rapid response to growth arrest and, by
virtue of its brief half-life, the ability to quickly resume growth as
changing conditions allow.
Diauxie
Experimental Design and Data
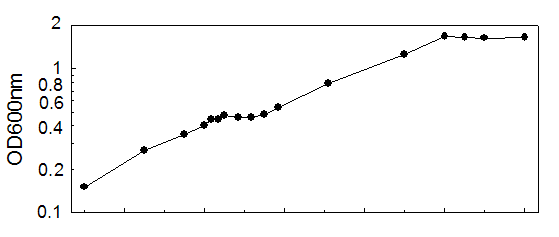
Time (min)
A600 Timepoint
90.0000 0.1500 logD1
135.0000 0.2700 logD2
165.0000 0.3500 logD3
180.0000 0.4020 logD4
185.0000 0.4400 logD5
190.0000 0.4400 logD
195.0000 0.4720 logD7
205.0000 0.4560 logD8
215.0000 0.4560 logD9
225.0000 0.4800 logD10
235.0000 0.5400 logD11
273.0000 0.7920 logD12
345.0000 1.4080 logD13
360.0000 1.6800 logD14
375.0000 1.6480 logD15
390.0000 1.6400 logD16
420.0000 1.6480 logD17
Spot-finding and quantitation. Image analysis software (ArrayVision, Imaging Research, Inc.) was used for spot-finding and quantitation of the E. coli Panorama arrays. The raw spot intensities were represented in a row-column format and exported into Microsoft Excel spreadsheets for further analysis, or as comma-delimited files (.csv) for upload to the database. Raw data from each experimental replicate were analyzed in Excel workbooks containing manually executed macros written in Visual Basic, or the data were processed in the database. The first step in the analysis associates the array coordinate for each spot with a unique spot number, the gene name, and related gene annotation. On the membrane arrays there are two spots for each gene, and these were treated as separate determinations. The raw data were normalized by expressing spot intensities as a percentage of the sum of all of the gene-specific spot intensities. The second step in the analysis applies the student t-test to determine the probability that the average of the experimental replicates is significantly different from the average of the control replicates . The P values (derived from the student t-test) for the normalized and natural log transformed data were calculated. The third step calculates relative gene expression between conditions by introducing a threshold value, chosen to be representative of the limit of detection of expressed genes (usually the 500th lowest expressed gene), and then calculating the ratio of the experimental/control expression levels such that genes that are more highly expressed in the experimental condition are given a positive value, and genes that are more highly expressed in the control condition are given a negative value.
Control for ratio calculation. The early log phase control values used for the diauxie experiment represent the average of 12 hybridizations: two each from time points 1 and 2 of the H2O2-treatment and time points 1 and 3 of the glucose-lactose diauxie and 4 from time point 2 of the glucose-lactose diauxie. This approach is possible because gene expression is essentially steady-state in early-logarithmic phase and because there is excellent correlation between these time points. The use of these control values allowed us to directly compare the H2O2-treatment and glucose-lactose diauxie.
E. coli Gene Expression Database (Oracle) Interface
H2O2 Treatment Experimental Design

100.0000 0.2480 logH1
140.0000 0.3840 logH2
145.0000 0.3760 logH3
150.0000 0.3800 logH4
160.0000 0.3960 logH5
170.0000 0.4080 logH6
180.0000 0.4160 logH7
200.0000 0.5040 logH8
255.0000 0.9000 logH9
330.0000 1.7800 logH10
345.0000 1.7840 logH11
390.0000 1.7840 logH12
Culture conditions. E. coli MG1655 (CGSC #6300) was cultured in 1 liter of morpholinepropanesulfonic acid (MOPS) minimal medium containing 2 g/liter of glucose (Neidhardt et al., 1974) in a 2 liter Biostat B fermentor. The temperature was maintained at 37 degrees C and pH was kept constant at 7.2 by the addition of 2 M NaOH. The dissolved oxygen level was maintained above 20% of its saturation level by adjusting the agitation speeds in the range of 400-600 rpm with fixed 1 liter/min air flow rate. Growth was monitored by measuring the optical density (OD) at 600nm. Immediately following timepoint 2 (H2), a sublethal concentration of H2O2 (50 ug/ml) was added to cause an immediate inhibition of growth, followed by a 40 min lag prior to resuming growth. The culture continued growing until entering stationary phase upon exhaustion of carbon and nitrogen.
E. coli Gene Expression Database (Oracle) Interface
Aerobic and Anaerobic Growth Experimental Design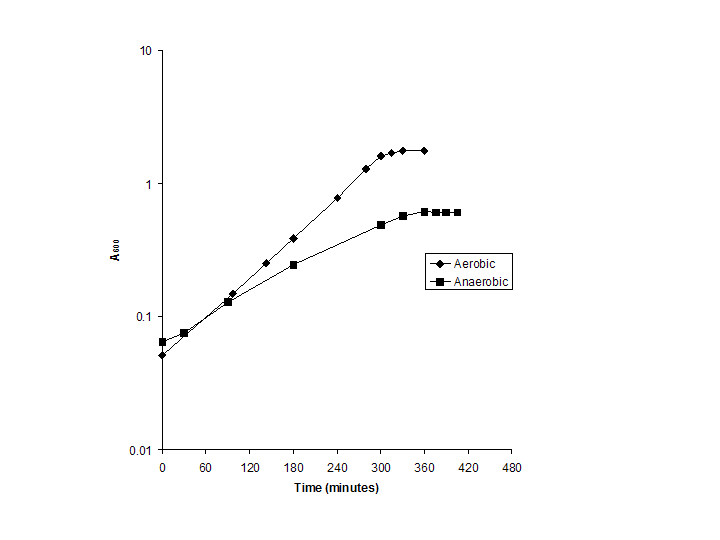
| Time (min) |
Aerobic |
Timepoint |
Time (min) |
Anaerobic |
| 0 |
0.051 |
1 |
0 |
0.065 |
| 97 |
0.148 |
2 |
30 |
0.0755 |
| 143 |
0.252 |
3 |
90 |
0.129 |
| 180 |
0.388 |
4 |
180 |
0.247 |
| 240 |
0.775 |
5 |
300 |
0.4905 |
| 280 |
1.285 |
6 |
330 |
0.568 |
| 300 |
1.61 |
7 |
360 |
0.613 |
| 315 |
1.7 |
8 |
375 |
0.6105 |
| 330 |
1.765 |
9 |
390 |
0.61 |
| 360 |
1.765 |
10 |
405 |
0.605 |
Experimental Design
Culture conditions. E. coli MG1655 (CGSC #6300) was cultured in 1 liter of morpholinepropanesulfonic acid (MOPS) minimal medium containing 2.0 g/liter of glucose (Neidhardt et al., 1974) in a 2 liter Biostat B fermentor. The temperature was maintained at 37 degrees C and pH was kept constant at 7.2 by the addition of 2 M NaOH. For the aerobic culture, the dissolved oxygen level was maintained above 20% of its saturation level by adjusting the agitation speeds in the range of 400-600 rpm with fixed 1 liter/min air flow rate. For the anaerobic culture, the medium contained 35 mM Na H2CO3 and was sparged with a mixture of nitrogen and carbon dioxide gases (90:10) at a fixed 1 liter/min flow rate. Growth was monitored by measuring the optical density (OD) at 600nm.
Data Legend
Aerobic
Growth Data Set (Timepoint 2 control; Excel: 3.1 Mb)
TGC_2_conpct: average normalized spot intensity for control
timepoint 2; values expressed as a percentage of the sum of all of the
gene-specific spot intensities
TGC_10_tstpct: average normalized spot intensity for
test timepoint 10
TGC_10_V_2_logratio: log10 ratio of timepoint 10 vs.
timepoint 2
TGC_10_V_2_pln: P value for the corresponding log ratio
calculated from the normalized, natural log transformed data
Aerobic
Growth Data Set (Timepoint 1-4 averaged control; Excel: 3.4 Mb)
TGC_1-4_conpct : average normalized spot intensity for
control timepoints 1-4; values expressed as a percentage of the sum of
all of the gene-specific spot intensities
TGC_1_tstpct: average normalized spot intensity for test
timepoint 1
TGC_1_logratio: log10 ratio of timepoint 1 vs. control
average timepoints 1-4
TGC_1_pln: P value for the corresponding log ratio calculated
from the normalized, natural log transformed data
AGTC_2_conpct: average normalized spot intensity for control timepoint 2; values expressed as a percentage of the sum of all of the gene-specific spot intensities
AGTC_10_tstpct: average normalized spot intensity for test timepoint 10
AGTC_10_V_2_logratio: log10 ratio of timepoint 10 vs. timepoint 2
AGTC_10_V_2_pln: P value for the corresponding log ratio calculated from the normalized, natural log transformed data
Anaerobic
Growth Data Set (Timepoint 1-4 averaged control; Excel: 3.4 Mb)
AGTC_1-4_conpct : average normalized spot intensity for
control timepoints 1-4; values expressed as a percentage of the sum of
all of the gene-specific spot intensities
AGTC_1_tstpct: average normalized spot intensity for
test timepoint 1
AGTC_1_logratio: log10 ratio of timepoint 1 vs. control
average timepoints 1-4
AGTC_1_pln: P value for the corresponding log ratio calculated
from the normalized, natural log transformed data
Aerobic
vs. Anaerobic Comparison (Timepoints 1-6 averaged Excel: 1.5Mb)
Aer_pct: average normalized spot intensity for aerobic
timepoints 1-6; values expressed as a percentage of the sum of all of
the gene-specific spot intensities
Anaer_pct: average normalized spot intensity for anaerobic
timepoints 1-6
Anaer/Aer_LOGRATIO: log10 ratio of Anaerobic average
timepoints 1-6 vs. Aerobic average timepoints 1-6
Anaer/Aer_PLN: P value for the corresponding log ratio
calculated from the normalized, natural log transformed data
Aer_STDDEV: Standard deviation of Aerobic timepoints
1-6
Anaer_STDDEV: Standard deviation of Anaerobic timepoints
1-6
E. coli Gene Expression Database (Oracle) Interface